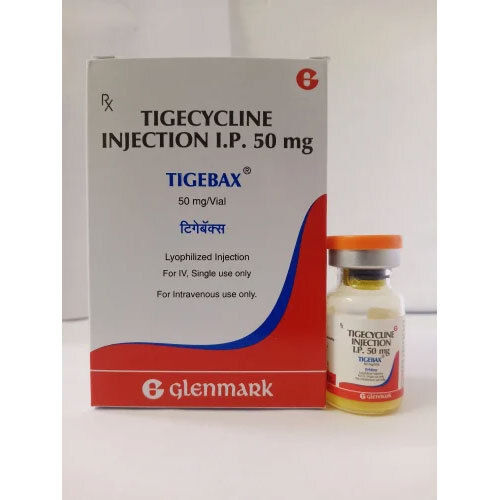
Tigecycline 50 Mg Injection
Product Details:
- Origin of Medicine Synthetic
- Assay ≥98%
- CAS No 220620-09-7
- Grade Pharmaceutical
- Storage Store in a refrigerator (2°C–8°C) and protect from light
- Indication Treatment of complicated skin and intra-abdominal infections as well as community-acquired bacterial pneumonia
- Medicine Type Antibiotic
- Click to view more
X
Tigecycline 50 Mg Injection Product Specifications
- C29H39N5O8
- Antibiotic
- ≥98%
- 220620-09-7
- Synthetic
- Treatment of complicated skin and intra-abdominal infections as well as community-acquired bacterial pneumonia
- Store in a refrigerator (2°C–8°C) and protect from light
- Pharmaceutical
- Tigecycline
- Administered intravenously for bacterial infections as prescribed by a physician
- 10 vials
- Yellow to orange lyophilized powder
- 2 years from the date of manufacture
- 50 mg
- Injection
Other Products in 'Antibiotic Medicine' category
Get in touch with us